Perils and Pitfalls of Obstetric Neuraxial Anaesthesia
Colleen Harnett1, Terry Tan2
1Fellow in High Risk Obstetric Anaesthesia, and Quality & Patient Safety, Coombe Hospital.
2Consultant Anaesthesiologist, Coombe Hospital
Introduction
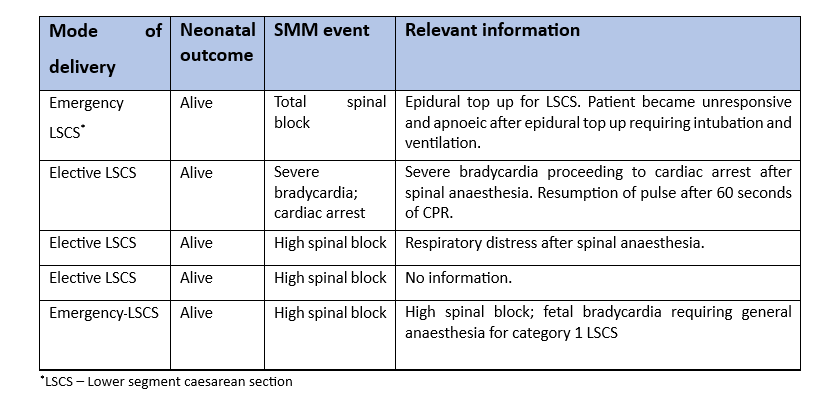
The National Perinatal Epidemiology Centre (NPEC) annual report on Severe Maternal Morbidity (SMM) 2021 identified 5 cases requiring ICU/CCU admission categorised under the term “anaesthetic complication” (Leitao et al., 2022). All 5 cases were related to neuraxial anaesthesia for caesarean section. There were 21,523 caesarean sections carried out in Ireland in 2021 giving an SMM incidence of 1:500,000 (0.0002%) due to anaesthesia complications. Although these incidents are extremely rare, it can be life threatening or cause catastrophic injury to mother and baby. A summary of the incidents is shown in the table below.
High/total spinal block
A ‘High spinal block’ is defined as a sensorimotor block higher than a level of T4. Initially a patient may present with altered sensation in the hands and/or a sensation of difficulty breathing. Cervical nerve root involvement (C6-T1) can be assessed by grip strength of the hands (Hadzic, 2017). For most patients, reassurance is adequate but for some there may be a rapidly ascending component with an ascending block of sympathetic, sensory and motor nerves as the block ascends to involve the cervical spinal resulting in dyspnoea, difficulty in phonation, bradycardia, and hypotension. In a ‘total spinal’ there may be a progression to loss of consciousness secondary to cerebral hypoperfusion or as the block spreads above the spinal cord to the brainstem (Hadzic, 2017; Skowronski & Rigg, 1981).
Incidence of high/total spinal block
The incidence of a high spinal/total spinal block is rare and is estimated to be 1: 4336 (D’Angelo et al., 2014). Pooled data from 14 units in the UK between 1987 to 2003 showed the incidence of total spinal block as a complication following epidural anaesthesia for labour to be far more uncommon at an estimated incidence of 1: 16200 (0.006%) (Jenkins, 2005). Although a rare occurrence, high/total spinal block is the leading cause of antepartum cardiac collapse in the UK. Beckett et al, 2017 reported that out of a total of 66 cardiac arrests that occurred in the obstetric setting between 2011 and 2014, 10 occurred followed total spinal anaesthesia (Beckett et al., 2017).
Prevention of high/spinal block
Total spinal anaesthesia can occur with an unrecognised or inadvertent accidental dural puncture during epidural catheter placement, followed by an unintentional administration of an epidural ‘top-up’ dose of local anaesthetic into the intrathecal space via the misplaced catheter (Hadzic, 2017). High spinal block can also occur when spinal anaesthesia is attempted following failed epidural anaesthesia. Management strategies include reducing the dose of spinal anaesthesia after a failed epidural top up for caesarean section, or not topping up a poorly functioning epidural catheter, but proceeding directly to a spinal anaesthetic (D’Angelo et al., 2014). Accidental subdural catheter placement resulting in a subdural block may account for delayed onset, profound and extensive blockade(Reynolds & Speedy, 1990). Early signs of a subdural block can manifest as ‘unexplained’ neurological sequelae such as inadequate ‘patchy’ blocks or delayed onset of extensive sensory block with minimal motor block. The catheter should be re-sited in such cases. Other factors that affect the distribution of local anaesthetic within the subarachnoid space include density of the local anaesthetic solution, patient position at the time of spinal administration, height of the patient, increased abdominal pressure (pregnancy and truncal obesity), and gestational age of pregnancy (Hadzic, 2017). All these factors should always be considered when administering spinal anaesthesia for the pregnant patient.
The epidural catheter should always be tested prior to top up to exclude intrathecal placement. Though there is no international consensus on how the epidural catheter should be tested, gentle aspiration followed by a test dose is an acceptable technique. Once the epidural catheter is placed, it should be aspirated with a 2ml syringe to visually confirm that no cerebral spinal fluid is withdrawn through the catheter. Negative aspiration does not negate the risk of an inadvertent intrathecal catheter and the onset of a motor block or a profound hypotension following a test dose administration of local anaesthetic medication should raise suspicion of intrathecal placement. Test doses should be sufficient to produce an obvious motor block but not cause a high block e.g. 10 mg of bupivacaine. Following these tests, it is prudent to give a loading dose of local anaesthetic with continued monitoring of blood pressure and ongoing motor assessment. Subdural catheters cannot be detected by negative aspiration test, or test dose. There is always a potential for the epidural catheter to migrate into the intrathecal or subdural space, and as such, there should be regular frequent monitoring of block height, and motor blockade of lower limbs while on pump maintenance of epidural analgesia for early detection.
Management of high/total spinal block
Management of high spinal block is largely supportive, waiting for the block to recede. Additional ventilatory support with high flow oxygen may be required. Respiratory compromise may require intubation and ventilation (Kar & Jenkins, 2001). Care should be taken to ensure the parturient is not aware during intubation with administration of an appropriate anaesthetic agent as the loss of respiratory effort and paralysis do not necessarily coincide with the loss of consciousness. There is frequently associated circulatory compromise and drugs that have a more stable haemodynamic profile such as ketamine, alfentanil, or midazolam (or combination) should be considered for induction of anaesthesia and prevention of awareness. After resuscitation, management is essentially supportive with continued ventilation with sedation until the block has adequately receded to a sufficient dermatomal level to facilitate extubation and return to normal ventilation.
Circulatory support with vagolytics, vasopressors, and intravenous fluids may be required to support maternal blood pressure and placental blood flow (Kar & Jenkins, 2001). Aorto-caval compression should be relieved with manual uterine displacement or by left lateral tilt. Urgent delivery may be required if there is foetal compromise.
Severe bradycardia after spinal anaesthesia for caesarean section
Hypotension is anticipated after spinal anaesthesia for caesarean section. In some cases, severe bradycardia leading to cardiac arrest can occur if venous return to the heart is inadequate because of aortocaval compression and can be further augmented by peripheral vasodilatation caused by sympathectomy. This phenomenon is described as the Bezold-Jarisch reflex when a bradycardic response of the heart is triggered by cardiac mechanoreceptor activation due to inadequate heart filling (Mark, 1983). Unopposed cardiac vagal activity after a spinal anaesthetic-induced sympathectomy further contributes to the bradycardia which can lead to asystole. Prevention is by applying a lateral tilt to the patient with a wedge and “co-loading” the patient with intravenous fluids after spinal anaesthesia. Bradycardia after spinal anaesthesia can be treated with the administration of atropine, and ephedrine to treat hypotension. Phenylephrine should not be used to treat hypotension associated with bradycardia due to the risk of asystole. In the event of a cardiac arrest, advanced cardiac life support should be commenced without delay including the immediate delivery of the foetus (Brown et al., 1972).
Conclusion
Neuraxial anaesthesia for caesarean section have multiple benefits over general anaesthesia such as reduced risk of aspiration, airway morbidity, risk of hypoxic brain damage, less neonatal depression, better birthing experience (awake for birth, skin to skin contact) and is the preferred technique of anaesthesia for caesarean section. However, neuraxial anaesthesia for caesarean section can also have severe complications and constant vigilance and awareness is required to ensure prevention, early detection, and management of complications to ensure optimal outcomes for mother and baby.
References
- Beckett, V., Knight, M., & Sharpe, P. (2017). The CAPS Study: incidence, management and outcomes of cardiac arrest in pregnancy in the UK: a prospective, descriptive study. BJOG: An International Journal of Obstetrics & Gynaecology, 124(9), 1374–1381. https://doi.org/10.1111/1471-0528.14521
- Brown, D., Carpenter, R., & Moore, D. (1972). Cardiac arrest during spinal anesthesia. . Anesthesiology, 36, 219–227.
- D’Angelo, R., Smiley, R. M., Riley, E. T., & Segal, S. (2014). Serious Complications Related to Obstetric Anesthesia. Anesthesiology, 120(6), 1505–1512. https://doi.org/10.1097/ALN.0000000000000253
- Hadzic, A. (2017). Hadzic’s Textbook of Regional Anesthesia and Acute Pain Management (2nd ed.). McGraw Hill/Medical.
- Jenkins, J. G. (2005). Some immediate serious complications of obstetric epidural analgesia and anaesthesia: a prospective study of 145 550 epidurals. International Journal of Obstetric Anesthesia, 14(1), 37–42. https://doi.org/10.1016/j.ijoa.2004.07.009
- Kar, G. S., & Jenkins, J. G. (2001). High spinal anaesthesia: two cases encountered in a survey of 81 322 obstetric epidurals. International Journal of Obstetric Anesthesia, 10(3), 189–191. https://doi.org/10.1054/ijoa.2001.0842
- Leitao, S., Manning, E., Corcoran, P., San Lazaro Campillo, I., & Keane, J. (2022). Severe Maternal Morbidity in Ireland Annual Report 2020.
- Mark, A. L. (1983). The Bezold-Jarisch reflex revisited: Clinical implications of inhibitory reflexes originating in the heart. Journal of the American College of Cardiology, 1(1), 90–102. https://doi.org/10.1016/S0735-1097(83)80014-X
- Reynolds, F., & Speedy, H. (1990). The subdural space: the third place to go astray. Anaesthesia, 45(2), 120–123. https://doi.org/10.1111/j.1365-2044.1990.tb14275.x
- Skowronski, G. A., & Rigg, J. R. A. (1981). Total Spinal Block Complicating Epidural Analgesia in Labour. Anaesthesia and Intensive Care, 9(3), 274–276. https://doi.org/10.1177/0310057X8100900312


